New coronavirus mutant strain detection reagent “E484Q mutation detection kit”
Fujifilm Wako Pure Chemical Industries, Ltd. (Headquarters: Chuo‐ku, Osaka, President: Kazuo Shiraki) will release the new coronavirus mutant strain detection reagent “E484Q Mutation Detection Kit” (research reagent) from today. This kit is one step RT‐qPCR method* 1By using it in, it is possible to detect the “E484Q” mutation of the Indian virus with high sensitivity.
* 1 Among the PCR methods, a method in which the reverse transcription reaction from RNA to DNA and the amplification reaction of DNA are performed in the same tube.
With the outbreak of the new coronavirus infection, mutant strains that are concerned about high infectivity and reduced vaccine efficacy are appearing one after another. Under these circumstances, Fujifilm Wako Pure Chemical Industries, Ltd. announced on March 29, this year, the “N501Y mutation detection kit” that detects the “N501Y” mutation of the British virus and the “N501Y mutation detection kit” that is common to South African and Brazilian viruses. Released the “E484K mutation detection kit” that can detect “E484K” mutations. In addition, on May 17, we started selling the “L452R mutation detection kit” that can detect the “L452R” mutation that is common to Indian and California viruses. I’m putting it in.
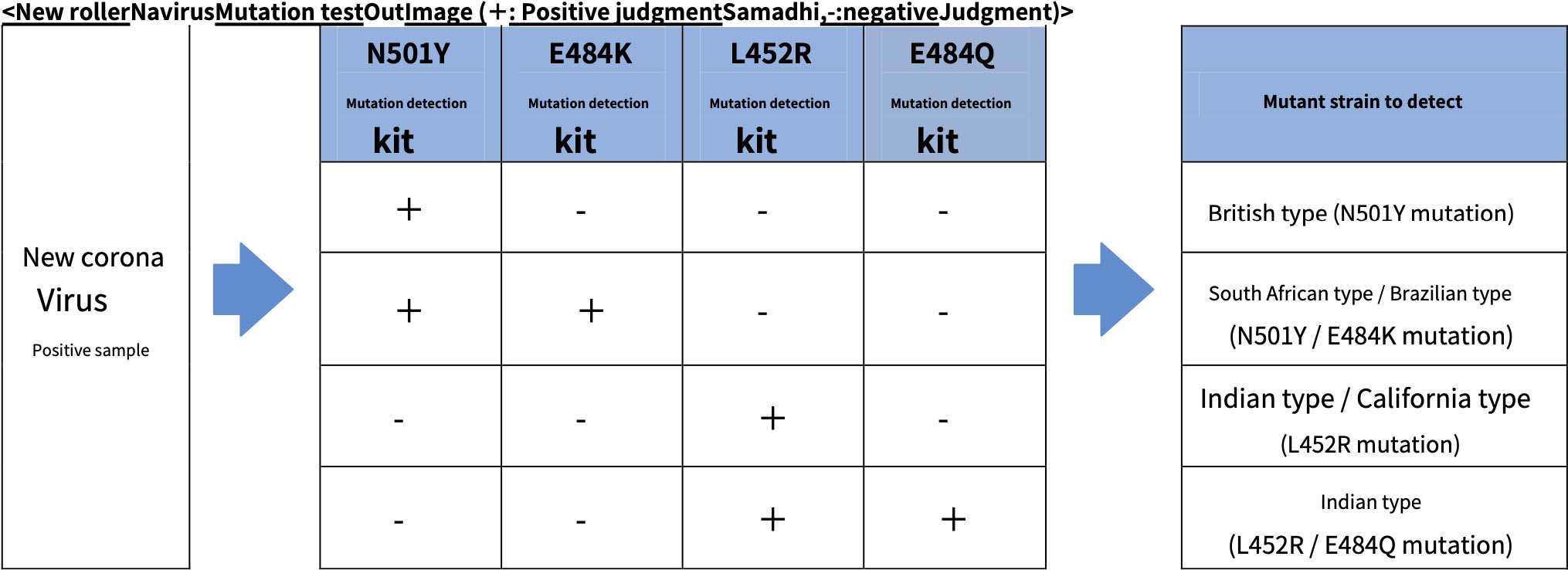
The “E484Q mutation detection kit” to be released this time is a reagent for detecting new coronavirus mutant strains developed by our original design method, and has a high “E484Q” mutation of Indian virus from positive samples of new coronavirus. It can be detected by sensitivity. Furthermore, in combination with the already released “L452R mutation detection kit”, it is possible to identify the type of Indian type virus that has two mutations, “L452R” and “E484Q”.
In addition to the “L452R mutation detection kit”, by using the “N501Y mutation detection kit” and “E484K mutation detection kit” together, (1) British type (N501Y), (2) South African type / Brazilian type (N501Y / E484K), and (3) It is possible to determine whether it corresponds to the Indian type / California type (L452R) or ➃ Indian type (L452R / E484Q).
We have developed a gene detection kit for the new coronavirus that realizes efficient PCR testing, and are providing it for applications such as administrative testing. Going forward, we will continue to promote research and development for swift response to new mutant strains, introduce reagents for detecting mutant strains to the market at an early stage, and strengthen the lineup to conduct a fact‐finding survey and infection of new coronavirus infections. We will contribute to the suppression of expansion.
Based on the philosophy of “becoming the power of the next science and creating the source of people’s happiness,” we will continue to develop and provide high‐ performance, high‐quality products that meet the needs of society and customers, and provide medical care. We will contribute to the development of industries and academic research in a wide range of fields, including.
Other Avigan/COVID-19/Hydro Ag +/Fujifilm Biomedical Coverage
(1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41)
Please don’t forget to follow us on Facebook, Twitter, Instagram, YouTube
Plus our owners’ groups
Fujifilm GFX Owners Group
Fujifilm X-H Owners Group
Fujifilm X-T Owners Group
Fujifilm X-S Owners Group
Fujifilm X-Pro Owners Group
Fujifilm X-E Owners Group
Fujifilm X-A Owners Group
Fujifilm X100 Owners Group
