Started Development of Highly Sensitive and Rapid Antigen Test Kit For New Coronavirus
Applying silver halide amplification technology for photo development
FUJIFILM Corporation (President: Kenji Sukeno) has started the development of a new coronavirus (SARS-CoV-2) antigen test kit. We aim to put the new coronavirus antigen test kit into practical use at an early stage by using the “silver amplification immunochromatography method * 1 ” that applies the silver salt amplification technology used in the photo development process, which enables quick and highly sensitive detection. In developing this test kit, we will receive an antibody that can detect the new coronavirus (SARS-CoV-2) antigen from Yokohama City University (Yokohama City, Kanagawa Prefecture).
The new coronavirus infection (COVID-19) was designated as a designated infectious disease in Japan on January 28, this year. In addition, on March 11, WHO confirmed that the spread of COVID-19 had reached a pandemic (a pandemic). According to the “Countermeasure Package” announced by the government on August 28, the average number of simple antigen tests per day is about 200,000 for the expansion of tests for the new coronavirus in preparation for the simultaneous epidemic with seasonal influenza. I will.
Antigen tests have the advantage of being able to perform tests quickly and confirming the results on the spot, but they are less sensitive than PCR tests, and higher sensitivity is required.
Since 2011, FUJIFILM has been selling “infectious disease rapid testing equipment” and special reagents for infectious diseases such as influenza using its own “silver amplification immunochromatography method”. It is used by many medical institutions as a highly sensitive antigen test system that can detect even when the amount of viruses and bacteria is small, such as in the early stages of development, and can obtain judgment results within a short time of 15 minutes.
Furthermore, in recent years, we have adapted the “Tuberculosis Rapid Diagnostic Kit” that can be visually inspected by applying the “Silver Amplification Immunochromatography Method” to European Pharmaceutical Affairs * 2 . High-sensitivity detection is achieved by using urine as a sample and combining a small amount of M. tuberculosis-specific components with a marker marker to generate large silver particles around it. Currently, research institutes in each country are conducting clinical evaluations to obtain WHO recommendations for supply to developing countries.
This time, we have confirmed that it is possible to increase the sensitivity of antigen testing by applying the “silver amplification immunochromatography method” to the antigen of the new coronavirus. In the future, we will proceed with the development of an antigen test kit that can quickly and highly sensitively test the presence or absence of the new coronavirus antigen in the sample.
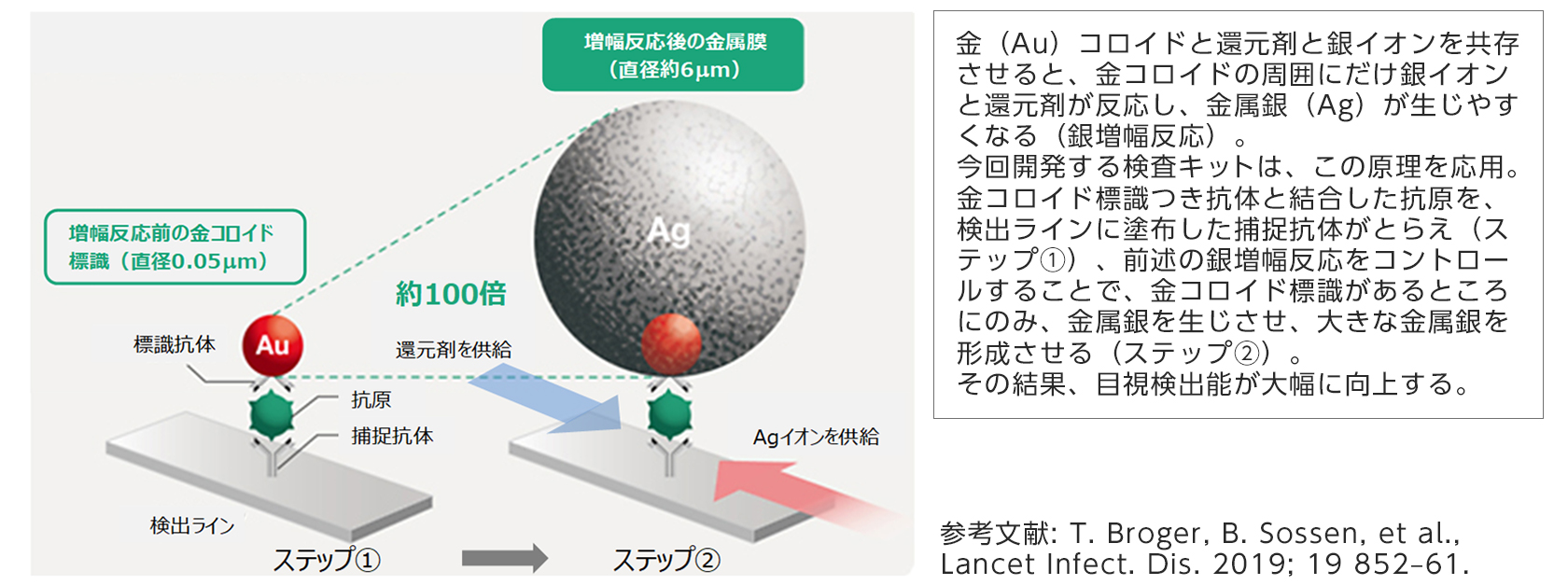
[Silver amplification technology applied to the antigen test kit developed this time]
The Fujifilm Group has supplied reagents for PCR testing and test reagents for the fully automated gene analyzer “Mutaswako g1” * 3 , which enables fully automatic, simple and rapid PCR testing, and has been testing new coronaviruses. Has contributed to the speeding up of. Going forward, we will continue to contribute to improving the efficiency of new coronavirus testing and improving the quality of medical care by providing a wide range of products and services that meet the needs of medical practice.
* 1 In the immunochromatography method, when a test substance (virus, etc.) is present in a sample (nasal swab, etc.) dropped into a reagent, it binds to the labeled antibody in the reagent to generate an antigen-antibody complex, and this complex is generated. A test method in which a colored line indicating positive (with antigen) is displayed when the body is captured by an antibody that is linearly applied on the detection line in advance. Since the results can be obtained easily and quickly, it is often used for testing for infectious diseases that require urgent treatment.
* 2 European Union (EU) Medical Device Directive for In Vitro Diagnostic 98/79 / EC (I VDD)
* 3 Medical device notification number 27B3X00024000016 Manufacturer / Distributor Fujifilm Wako Pure Chemical Industries, Ltd.
Other Avigan/COVID-19/Fujifilm Biomedical Coverage ( 1, 2, 3, 4, 5, 6, 7, 8, 9,10. 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26)
Please don’t forget to follow us on Facebook, Twitter, Instagram, YouTube
Plus our owners’ groups
Fujifilm GFX Owners Group
Fujifilm X-H Owners Group
Fujifilm X-T Owners Group
Fujifilm X-Pro Owners Group
Fujifilm X-E Owners Group
Fujifilm X-A Owners Group
Fujifilm X100 Owners Group
