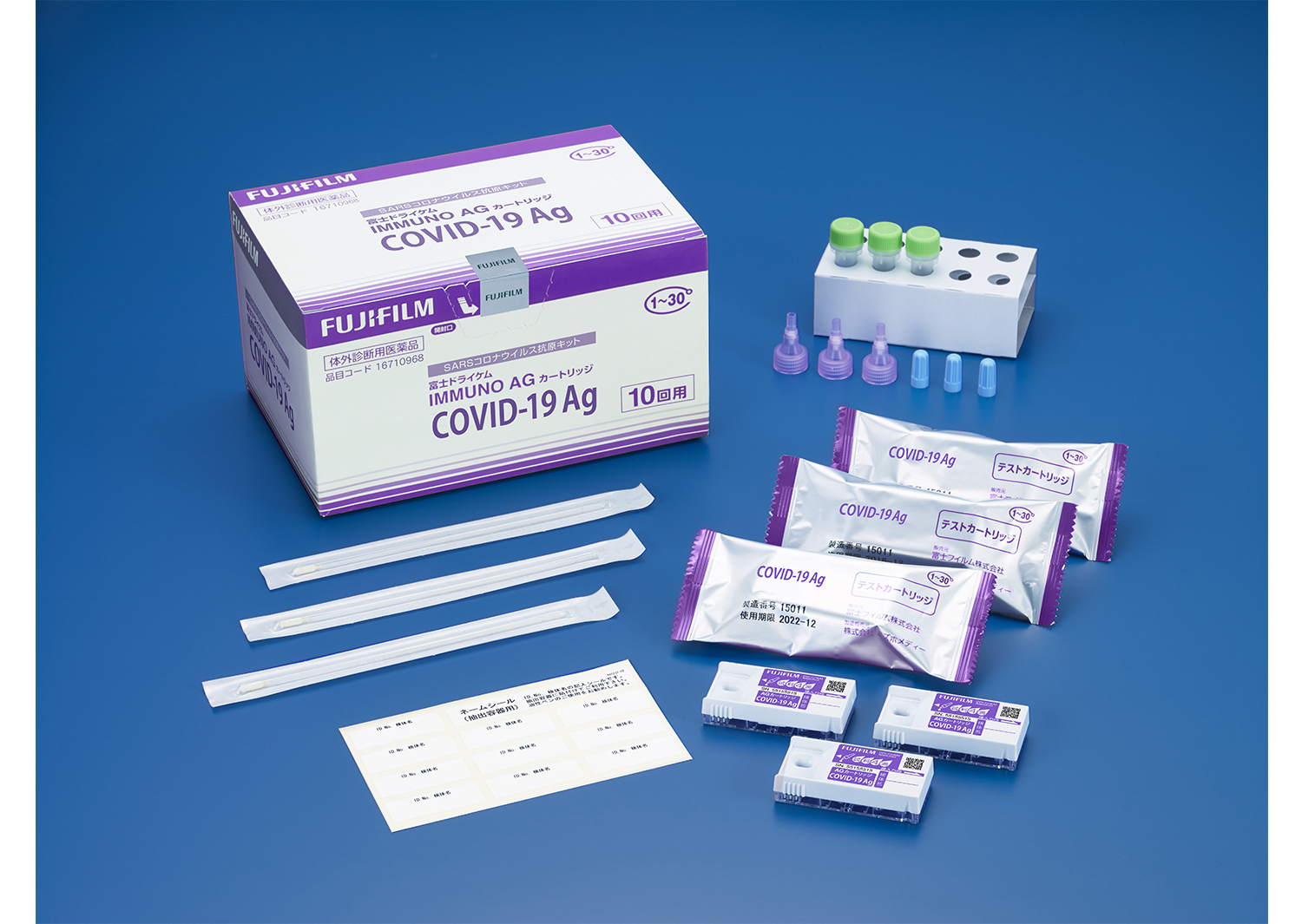
New release of new coronavirus antigen test kit “COVID-19 Ag”
Applying high-sensitivity detection technology by silver salt amplification reaction For infectious disease testing device “Fuji Dry Chem IMMUNO AG” series
FUJIFILM Corporation (President: Kenji Sukeno) has announced a new coronavirus (SARS-CoV-2) antigen test kit “Fuji Dry Chem IMMUNO AG” that applies high-sensitivity detection technology based on the silver salt amplification reaction used in the photo development process. In vitro diagnosis of the cartridge COVID-19 Ag (hereinafter, “COVID-19 Ag”) for our infectious disease testing device “Fuji Dry Chem IMMUNO AG” (hereinafter, IMMUNO AG) series. It will be released as a pharmaceutical product through FUJIFILM Medical Co., Ltd. (President: Yoshihiro Kawahara) from March 9th.
With the spread of the new coronavirus infection (COVID-19), the scope of antigen qualitative testing has been expanded in Japan from January 22, 2021. As a result, the antigen qualitative test, which was previously defined as 2 to 9 days after onset, is now possible from 1 to 9 days after onset. In addition, when it is difficult to carry out a PCR test in an area where infection has spread, it is effective to carry out a wide range of antigen qualitative tests at medical institutions and facilities for the elderly, which are often at high risk of aggravation, from the viewpoint of preventing the spread of infection. Since the Ministry of Health, Labor and Welfare has indicated that, it has become possible to carry out qualitative antigen tests as an administrative test for staff of medical institutions and facilities for the elderly, inpatients and residents, even if they are asymptomatic. * 1 . In the future, it is expected that antigen qualitative tests, which can be easily tested, will be performed in more situations, but in general, their sensitivity was lower than that of PCR tests, so higher sensitivity was required.
The “COVID-19 Ag” to be released this time uses Fujifilm’s original “silver amplification immunochromatography method * 2 ” to amplify the antigen label used in the normal immunochromatography method to a size about 100 times larger. It enables detection with a small amount of virus. In addition to the nasopharyngeal swab, the nasal swab can be collected and used by the subject himself under the control of the medical staff, which contributes to reducing the risk of infection by the medical staff.
The measurement is automatically started by simply dropping the sample extract onto the “COVID-19 Ag” and setting it in the infectious disease testing device “IMMUNO AG” series, and the judgment result can be obtained in 15 minutes. .. More than 25,000 units of the “IMMUNO AG” series have been introduced mainly in hospitals and clinics in Japan, and many facilities will be able to carry out simple new corona antigen tests using “COVID-19 Ag”. Furthermore, if you use the influenza virus antigen test kit for the “IMMUNO AG” series, you can test the antigens of the new corona virus and influenza virus from the same extract, so you can use multiple samples even when differentiation from influenza is required. There is no need to collect.
The Fujifilm Group has supplied reagents for PCR testing and test reagents for the fully automatic gene analyzer “Mutaswako g1” * 3 , which enables fully automatic, simple and rapid PCR testing, and has been testing new coronaviruses. Has contributed to the speeding up of. Going forward, we will continue to contribute to improving the efficiency of new coronavirus testing and improving the quality of medical care by providing a wide range of products and services that meet the needs of medical practice.
* 1 From the Ministry of Health, Labor and Welfare office communication “Guidelines for pathogen testing for new coronavirus infection (COVID-19) (3rd edition)” and how to carry out qualitative antigen testing (January 22, 2021). There are certain conditions for conducting antigen qualitative tests on medical institutions, staff of facilities for the elderly, and residents.
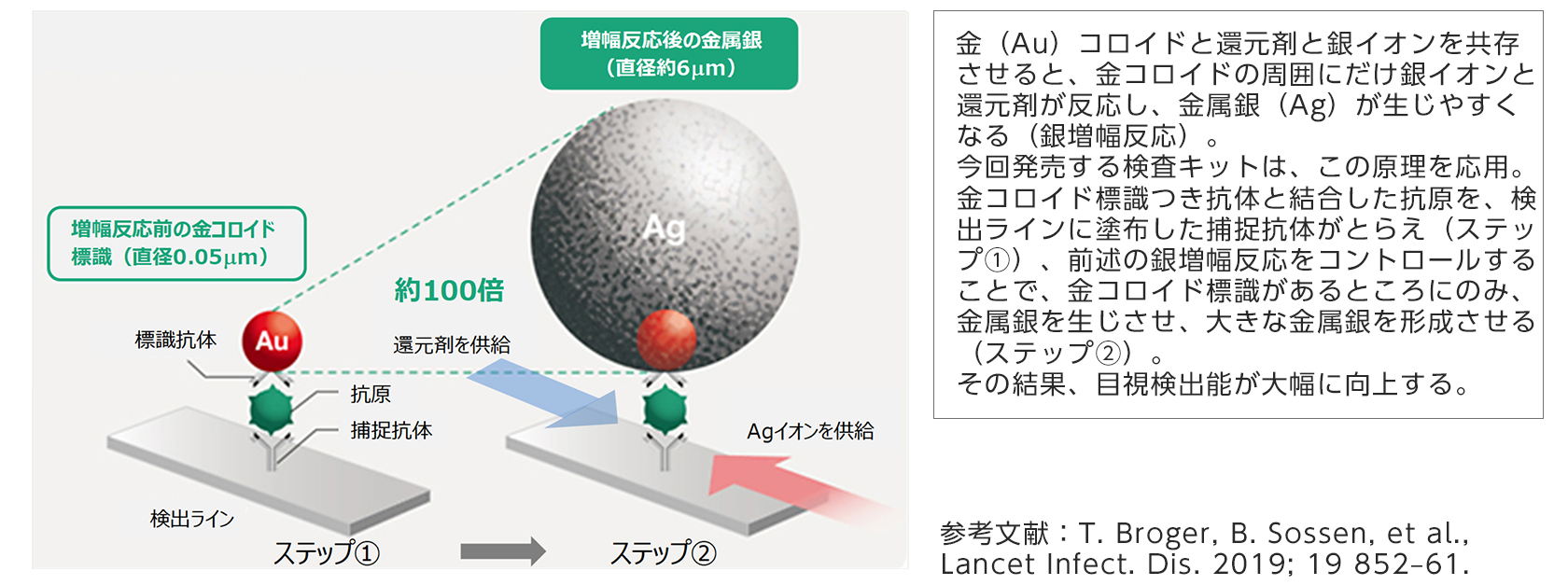
* 2 In the immunochromatography method, when a test substance (virus, etc.) is present in a sample (nasopharyngeal swab, etc.) dropped into a reagent, it binds to the labeled antibody in the reagent to generate an antigen-antibody complex. A test method in which a colored line indicating positive (with antigen) is displayed when the complex is captured by an antibody that is linearly applied on the detection line in advance. Since the results can be obtained easily and quickly, it is often used for testing for infectious diseases that require urgent treatment.
* 3 Medical device notification number 27B3X00024000016 Manufacturer / Distributor Fujifilm Wako Pure Chemical Industries, Ltd.
[Silver amplification technology applied to the antigen test kit to be released this time]
 Record
Record
1. Product name
SARS Coronavirus Antigen Kit
(Brand name: Fuji Drychem IMMUNO AG Cartridge COVID-19 Ag / Approval number: 30300EZX00024000)
<System that can use this kit>
Infectious disease testing device
(brand name: Fuji Dry Chem IMMUNO AG1 / notification number: 14B2X10002000104)
( brand name: Fuji Dry Chem IMMUNO AG2 / notification number: 14B1X10022000123)
( brand name: Fuji Dry Chem IMMUNO AG2 / Notification number: 14B2X10002000123)
2. Release date
March 9, 2021
3. Main features
(1) High sensitivity is achieved by applying high-sensitivity detection technology by silver salt amplification reaction.
By using photographic film development technology, the label that serves as an antigen marker is amplified about 100 times compared to the case without this technology to improve visibility and sensitivity, even when the amount of virus is small. It can be detected.
(2) Antigen testing for new coronavirus and influenza virus is possible from the same sample
When differentiation from influenza is required, both diseases can be tested by applying the sample extract to the antigen test kits for the new coronavirus and influenza virus.
(3) Not only nasopharyngeal swab, but also nasal swab can be used as a sample.
In addition to the nasopharyngeal swab, the sample can also be a nasal swab collected by the subject under the control of the medical staff, which contributes to reducing the risk of infection for the medical staff.
(4) Easy inspection is possible with the “Fuji Dry Chem IMMUNO AG” series, which has been installed in more than 25,000 units in Japan.
When the “COVID-19 Ag” in which the subject’s sample is dropped is set in the “Fuji Dry Chem IMMUNO AG” series of infectious disease testing devices that are widely used in domestic medical institutions and clinics, the measurement starts automatically for 15 minutes. You can get the judgment result with.
4. Main specifications
product name
SARS Coronavirus Antigen Kit
“Fuji Dry Chem IMMUNO AG Cartridge COVID-19 Ag”
Measurement item
New coronavirus (SARS-CoV-2 antigen)
Specimen
Nasopharyngeal swab, nasal swab
Reaction time
15 minutes
Storage method
Room temperature (1-30 ° C)
Validity period
12 months
Sales unit
10 times / box
Composition
Test cartridge, extract (0.6 mL), sterile cotton swab (for nasopharyngeal swab), filter (for extract), blue cap (for primary storage of extract): 10 each, stand (for extract), name sticker :Each one

Other Avigan/COVID-19/Hydro Ag +/Fujifilm Biomedical Coverage (1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38)
Please don’t forget to follow us on Facebook, Twitter, Instagram, YouTube
Plus our owners’ groups
Fujifilm GFX Owners Group
Fujifilm X-H Owners Group
Fujifilm X-T Owners Group
Fujifilm X-S Owners Group
Fujifilm X-Pro Owners Group
Fujifilm X-E Owners Group
Fujifilm X-A Owners Group
Fujifilm X100 Owners Group
