- New Coronavirus N501Y Mutation Detection Kit
- New Coronavirus E484K Mutation Detection Kit
High-sensitivity detection of mutant new coronavirus SARS-CoV-2 New coronavirus “N501Y” “E484K” mutation detection kit released British, South African, and Brazilian viruses detected (Fujifilm Wako Junyaku)
Fujifilm Wako Pure Chemical Industries, Ltd. (Headquarters: Chuo‐ku, Osaka, President: Kazuo Shiraki) is a one‐step RT‐qPCR.
Law * The “N501Y Mutation Detection Kit” and “E484K Mutation Detection Kit” (both research reagents) that detect mutant new coronavirus (SARS‐CoV‐2) with high sensitivity will be released today.
The new coronavirus, whose infection is spreading worldwide, has been confirmed to have multiple mutant viruses in each country. There are concerns about its infectivity and its relationship with the effectiveness of vaccines. Especially in Japan, England and South Africa And the Brazilian type is gradually expanding. These mutant viruses are highly infectious and reinfectious It has been pointed out that the risk of infection may increase and the effectiveness of the vaccine may decrease. Containment is required.
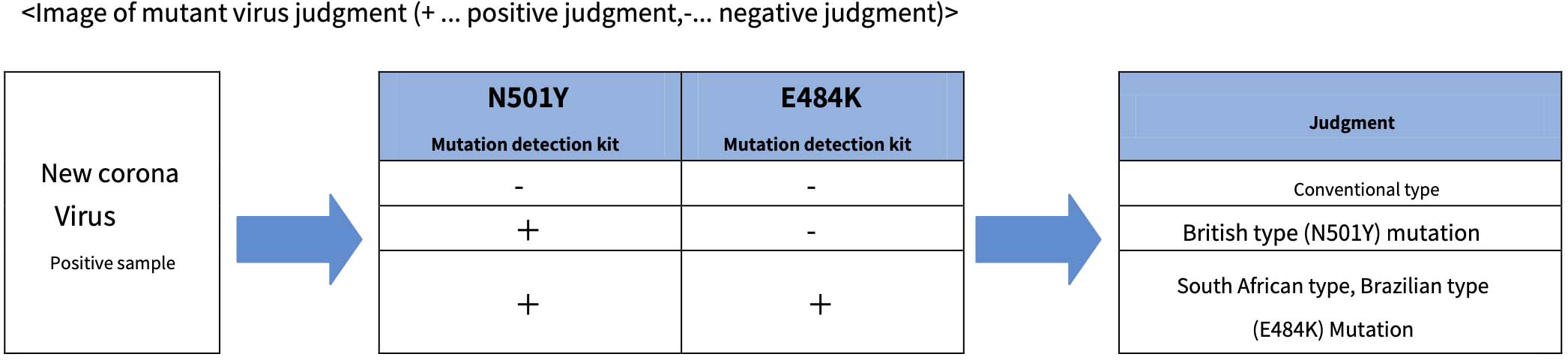
The “N501Y mutation detection kit” and “E484K mutation detection kit” to be released this time are genetic test kits that detect mutant new coronaviruses using the one‐step RT‐qPCR method. “N501Y Mutation Detection Kit” is a British type
Detects genes that are common to multiple mutant viruses, including. “E484K mutation detection kit” excludes British type Detects genes common to South African and Brazilian mutant viruses. Positive for new coronavirus
By using the “N501Y mutation detection kit” and “E484K mutation detection kit” for RNA extracted from a sample, it is possible to determine whether it corresponds to the conventional type, the British type, the South African type, or the Brazilian type.
It is said that this kit is difficult to detect by the one‐step RT‐qPCR method by using our original design method. E484K gene mutations that are present can also be easily detected. This technology can be applied to detect various mutant viruses. Yes, even if a new mutant virus emerges in the future, we can respond quickly.
We have been selling a gene detection kit for the new coronavirus since April 2020, and it is used for administrative tests. Going forward, our corporate philosophy of “becoming the power of the next science and creating a source of happiness for people” will continue.
We will contribute to the fact‐finding survey of new coronavirus infections and measures to prevent the spread of infections.
* A DNA amplification reaction that uses RNA as a template, in which a reverse transcription reaction and a DNA amplification reaction are continuously performed in the same tube.
Other Avigan/COVID-19/Hydro Ag +/Fujifilm Biomedical Coverage
(1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39)
Please don’t forget to follow us on Facebook, Twitter, Instagram, YouTube
Plus our owners’ groups
Fujifilm GFX Owners Group
Fujifilm X-H Owners Group
Fujifilm X-T Owners Group
Fujifilm X-S Owners Group
Fujifilm X-Pro Owners Group
Fujifilm X-E Owners Group
Fujifilm X-A Owners Group
Fujifilm X100 Owners Group